Intravenous ibuprofen versus sodium valproate in acute migraine attacks in the emergency department: A randomized clinical trial
a b s t r a c t
Objective: Sodium valproate and ibuprofen are drugs with known efficacy in the treatment of headache associ- ated with acute migraine attacks. In this study, our aim was to compare the efficacy of these two drugs in the treatment of acute migraine attacks when administered as a single intravenous (IV) dose in the emergency de- partment.
Materials and method: This study was designed as a prospective, randomized controlled, double-blinded study and included patients aged 18 to 65 years who presented to the emergency department with acute headache and met the criteria of ‘migraine without aura’ according to the International Classification of Headache Disor- ders. The patients were randomized into two groups and given a single dose of 800 mg sodium valproate or 800 mg ibuprofen in 150 mL of normal saline by IV infusion over five minutes. Changes in pain levels were assessed using the numerical rating scale (NRS) for pain over a two-hour period.
Results: Ninety-nine patients (49 patients in the sodium valproate group and 50 in the ibuprofen group) completed the trial, and their data were included in the statistical analysis. The mean decrease in the post- treatment delta NRS values was statistically significantly higher in the sodium valproate group than in the ibuprofen group. The mean differences were 1.69 [confidence interval (CI): 1.02-2.37, p<0.001], the mean differ- ence between N0 and N2 was 3.61 (CI: 2.96-4.26, p < 0.001), the mean difference between N0 and N3 was 4.11 (CI: 3.54-4.67, p < 0.001), and the mean difference between N0 and N4 was 3.92 (CI: 3.67-4.46, p < 0.001). The number of patients who achieved the primary endpoint of pain relief was significantly higher in the sodium valproate group than in the ibuprofen group (p < 0.001). According to the Kaplan-Meier analysis showing the rates of reaching the targeted endpoint, there was a significant difference in the efficacy of the two-treatment group (?2 = 79.98, CI: 80.35-99.65; p = 0.000).
(C) 2022
Patients presenting with acute migraine constitute more than half of primary headache cases evaluated in the emergency department [1]. According to the World Health Organization, migraine is the third most common disease in the world and the second most common neu- rological disorder in terms of the frequency of disability [2]. The Global Burden of Disease study reported that migraine ranked first among the causes of years of healthy life lost to disability for the 15-49 years group [3]. migraine headaches may appear to be innocent at first since they do
* Corresponding author at: Kahramanmaras Sutcu Imam Universitesi Tip Fakultesi Acil Tip Anabilim Dali, Onikisubat, Kahramanmaras, Turkey.
E-mail address: [email protected] (I. Akbas).
not pose a life-threatening risk. However, considering their impact on both the quality of life and the patient intensity of healthcare institu- tions, it is indisputable that they constitute an important Public health problem.
The pathogenesis of migraine has not yet been fully elucidated. In scientific literature, it has been suggested that migraine may originate from the association of the trigeminal nerve and its extensions with the intracranial vascular system [4]. It is also considered to be caused by the dysfunction of neurons in the central nervous system, especially in the brain stem and diencephalon [5]. With the identification of signal molecules that play a role in the formation of migraine attacks, it has be- come easier to develop mechanism-basED treatments for the disease [6]. However, this situation is different in acute migraine attacks. In
https://doi.org/10.1016/j.ajem.2022.02.046
0735-6757/(C) 2022
these cases, emergency departments, to which patients with migraine attacks most refer, aim to relieve pain quickly and effectively. Different strategies are used in the treatment of acute attacks. Drugs to be used in the treatment process are classified into two categories as non- specific and specific [7]. Non-steroidal anti-inflammatory drugs (NSAIDs), acetaminophen, aspirin and drugs targeting accompanying symptoms (antiemetics, antihistamines, myorelaxants, and antiepilep- tics) are used as non-specific treatment options, while triptans and di- hydroergotamine are migraine-specific treatment agents.
NSAIDs are preferred as first-line therapy in the treatment of acute
migraine attacks, especially in emergency departments [8]. Ibuprofen, an NSAID, is an effective analgesic that can be used in the treatment of acute migraine according to the guidelines of the American Headache Society (AHS) and the American Academy of Neurology [9].
Sodium valproate is an antiepileptic that has been used orally for many years in migraine prophylaxis with proven efficacy [10]. Studies on the use of sodium valproate intravenously (IV) in the treatment of acute migraine attacks started relatively later. Research concerning the use of this drug has yielded heterogeneous results in terms of Analgesic efficacy [2]. When evaluated in terms of safety, there are positive results regarding the use of sodium valproate in acute migraine attacks [11,12]. To date, many randomized controlled clinical studies have been con- ducted to examine the efficacy of sodium valproate; however, it remains in the category of drugs with Level C strength of evidence in the treat- ment of acute migraine in the AHS guidelines [1].
Migraine analgesia is a multi-faceted clinical condition that cannot be limited to a single medical treatment. Expecting that every patient presenting with an acute attack will benefit from the same medical treatment will cause re-admissions due to insufficient analgesia and lead to the loss of workforce. In this study, our aim was to compare the analgesic efficacy of a single dose of IV sodium valproate and a single dose of IV ibuprofen in acute migraine attacks. We evaluated IV sodium valproate in order to seek a drug that can occasionally be used as an al- ternative to frequently administered medical treatments with known efficacy in clinical practice and compare the efficacy of different options.
This study was designed as a prospective, randomized controlled, double-blinded study. Patients aged 18 to 65 years who presented to the emergency departments of the hospitals where the study was con- ducted with a headache between January 1 and August 1, 2021 were evaluated for their eligibility for inclusion in the study. Ethical approval was obtained from the Ethics Committee of Ataturk University Faculty of Medicine, which was one of the participant centers.
The study included patients with a known diagnosis of migraine or those that met the criteria of ‘migraine without aura’ according to the International Classification of Headache Disorders, third edition (ICHD-3) [13]. All the patients were informed about the study proce- dures. Written consent was obtained from the volunteers who agreed to participate in the study.
Exclusion criteria
- Clinical suspicion of secondary or organic headache
- Hemodynamic instability
- Use of painkillers within the last six hours before presentation
- Regular use of analgesics (due to conditions such as chronic pain syn- drome, rheumatoid arthritis, and osteoarthritis) or and/or sodium valproate (due to epilepsy)
- Allergy to NSAIDs and narcotic analgesics
- Pregnancy or suspected pregnancy
- Chronic and uncontrolled heart failure, history of gastrointestinal bleeding, chronic kidney failure, chronic Liver failure, and history of epilepsy
- Intervention
According to their treatment groups, the patients were administered 800 mg of sodium valproate (Depakin, Sanofi-Aventis, France 400 mg/4 mL) or 800 mg of ibuprofen (Intrafen, GEN Pharmaceuticals, Turkey 800 mg/8 mL). In both treatment groups, the drugs were administered in 150 mL of normal saline by IV infusion over five minutes.
Randomization was performed by the medical specialist who planned the study, using an online random enumerator. Then, the same person prepared the drug mixtures to be administered and placed them in treatment boxes numbered in the same order as in randomiza- tion. The drugs prepared for the treatment groups had similar appear- ance. Therefore, the doctor who evaluated the patient and arranged the analgesic treatment and the nurse who administered the drug were blinded to the groups of the patients. It was also planned to admin- ister IV fentanyl at a dose of 1 ug/kg to the patients as rescue treatment when necessary.
-
- Outcomes
The Numerical Rating Scale for pain was used to evaluate pain levels starting from the presentation of the patients. The patients were asked to describe their pain by rating it from 0 (no pain) to 10 (worst pain ever experienced) [14]. The NRS score at the time of presentation to the emergency department was recorded during the triage and noted as N0. It was aimed to initiate IV analgesic therapy within a max- imum of 10 min in each patient. After treatment, the NRS scores were recorded at minutes 30, 60, 90 and 120 as N1, N2, N3 and N4, respec- tively. The mean changes (delta NRS) that occurred during the treat- ment process and their percentages were also noted. The mean percent decrease was calculated using the following formula: (N0 – N1
/ N0) x 100. The primary endpoint of the study, pain relief, was deter- mined based on a decrease of >=50% in the post-treatment NRS score compared to the baseline [15]. The variables evaluated in the secondary outcome targets were requirement of rescue treatment at least once during the study and recurrence of attacks after discharge. The distribu- tion of the patients with these characteristics was examined according to the treatment groups. In order to evaluate the recurrence of attacks, the patients were contacted by phone at the 48th hour after discharge and asked whether they had another migraine attack.
-
- Statistical analysis
The Statistical Package for the Social Sciences (SPSS, version 20.0) (IBM Corp., Armonk, NY, USA) was used for the statistical analysis of the data obtained from the study. Continuous variables conforming to the normal distribution were expressed as mean +- standard deviation (SD), and categorical variables were given as numbers and percentages. The unpaired t-test (for normally distributed data) and the chi-square test were used when comparing the demographic data, baseline charac- teristics, mean differences in the NRS scores, and frequency of side effects between the treatment groups. The Kaplan-Meier survival anal- ysis and the log-rank test were conducted to estimate the time to pain relief and compare treatment success between the two treatment groups over the two-hour period. The 95% confidence interval (CI) was used to express the data. Test results with a p value below 0.05 were considered statistically significant.
-
- Sample size
When calculating the sample size, we referred to the literature com- paring sodium valproate with different comparators. In a previous study
in which the NRS score was evaluated, the mean NRS scores of the treat- ment groups with significant statistical results were calculated as 2.8 and 4.7, and the SD value as 2.68 [16]. In the sample size analysis we performed in line with these results, we calculated the Power of the study as 0.90 when 42 patients were included in each group (Type I error = 0.05).
- Results
- Patients
After applying the exclusion criteria, 112 patients were initially in- cluded in the study. Among these patients, the data of 13 patients
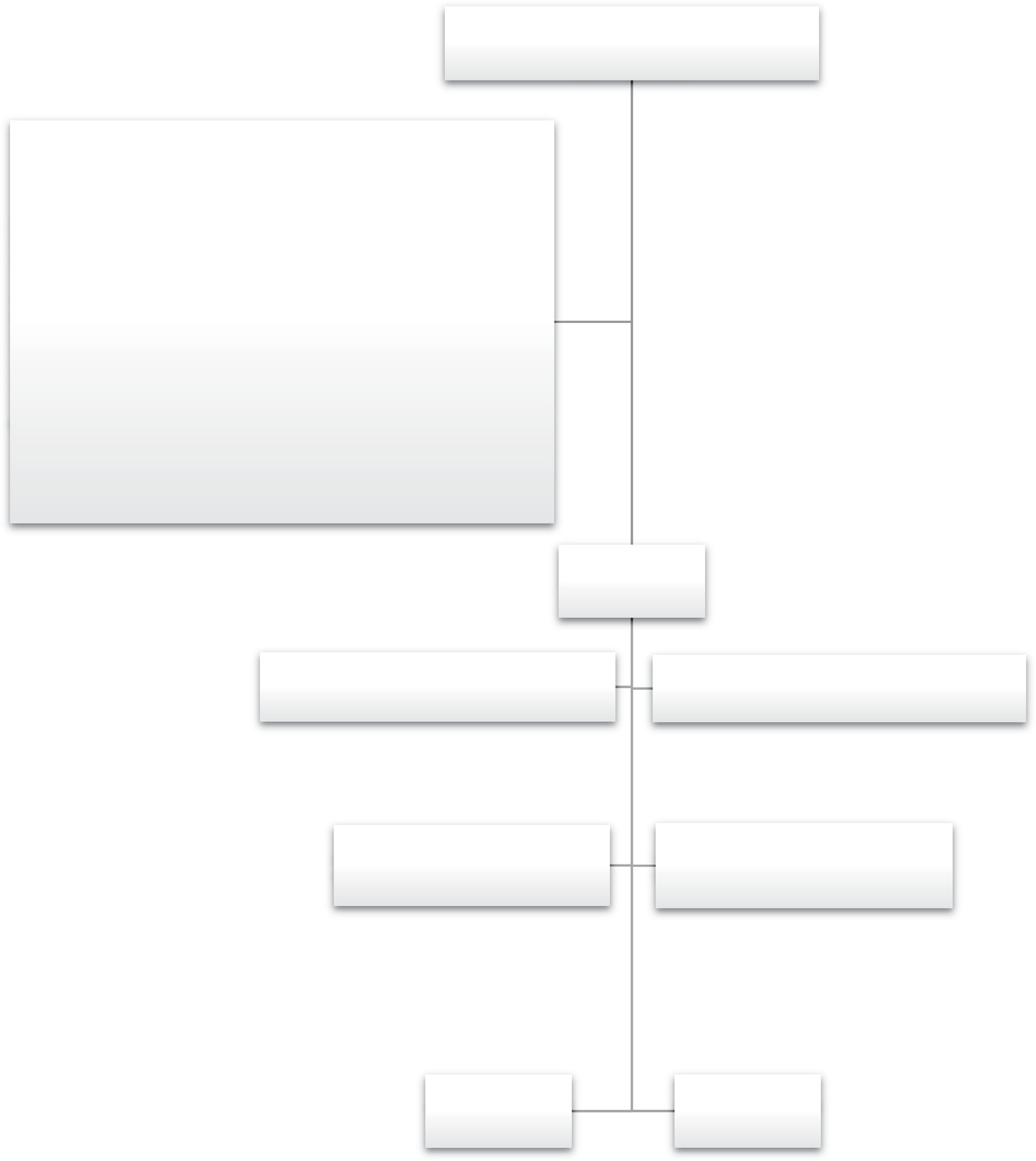 Assessed for eligibility
Assessed for eligibility
(n = 3093)
Excluded (n = 877):
Suspected secondary and/or organic headache (n = 51) Headache duration > 6 hours (n = 74)
Vital instability (n = 213) chronic conditions (n = 108) Allergic history (n = 26)
<18 years (n = 346) Pregnancy/lactation (n = 38) Unable to give consent (n = 9) Declined to participate (n = 12)
Did not meet the criteria for migraine without aura (n = 2104)
Randomized (n = 112)
Allocated to the IV Ibuprofen group (n = 56) Received medication (n = 56)
Allocated to the IV Sodium VA group (n = 56) Received medication (n = 56)
Lost to follow-up (n = 2) Discontinued intervention (n = 4)
Lost to follow-up (n = 5) Discontinued intervention (n = 2)
Completed trial; included in analysis (n = 50)
Completed trial; included in analysis (n = 49)
Fig. 1. Consolidated standards of reporting trials diagram of the study.
were excluded (six did not complete the study and seven could not be reached by phone). As a result, the study was completed with 49 pa- tients in the sodium valproate group and 50 in the ibuprofen group, and the data of these patients were analyzed. Fig. 1 presents the Consol- idated Standards of Reporting Trials diagram of the study.
The mean age of the 99 patients who completed the study was 34.7 (SD, 9.7; range, 18-55) years. The distribution of these patients by sex was 53 (53.5%) females and 46 (46.5%) males. There was no significant difference between the treatment groups in terms of the mean age and sex distribution (p > 0.05). Patient demographics, vital parameters, and clinical features of headache are shown in Table 1. When the treatment groups were compared in terms of the distribution frequency and mean values of the variables presented in this table, no statistically significant difference was found (p > 0.005).
-
- Efficacy
The mean NRS scores at the time of presentation were 9.0 +- 1.1 for the sodium valproate group and 8.9 +- 1.2 for the ibuprofen group, with no significant difference (p > 0.05). Fig. 2 was created to show the mean NRS changes for each measurement time. The mean differences be- tween the delta NRS values were as follows: 1.69 between N0 and N1 [confidence interval (CI): 1.02-2.37, p < 0.001], 3.61 between N0 and N2 (CI: 2.96-4.26, p < 0.001), 4.11 between N0 and N3 (CI: 3.54-4.67, p < 0.001), and 3.92 between N0 and N4 (CI: 3.67-4.46, p < 0.001). In
light of these results, the mean post-treatment decrease in the NRS score was statistically significantly higher in the sodium valproate group than in the ibuprofen group. When we examined the percent de- crease rates in the NRS values, the mean differences between the evalu- ation times were as follows: 18.9 between N0 and N1 (CI: 11.7-26.1, p < 0.001), 39.1 between N0 and N2 (CI: 32.9-45.3, p < 0.001), 44.0 be-
tween N0 and N3 (CI: 38.8-49.2, p < 0.001), and 41.9 between N0 and N4 (CI: 36.9-46.8, p < 0.001). The mean percent decrease in the NRS scores observed throughout the study was statistically significantly higher in the sodium valproate group than in the ibuprofen group. The number of patients who achieved the primary endpoint of our study, pain relief, was significantly higher than in the sodium valproate group (n = 49) compared to the ibuprofen group (n = 30) (p < 0.001). Fig. 3 presents the Kaplan-Meier curve showing the rate of reaching the endpoint at NRS score measurement times according to
Baseline demographic characteristics of patients according to treatment groups.
the treatment groups. The log-rank test revealed a significant difference in pain relief efficacy between the treatment groups (?2 value = 79.98, CI: 80.35-99.65; p = 0.000).
The total number of patients who required rescue analgesic treat- ment at least once during the study was 12, of whom five (41.7%) were in the sodium valproate group and seven (58.3%) in the ibuprofen group. There was no significant difference between the treatment groups in terms of the distribution of these patients (p = 0.563). The number of patients who had recurrent MIgraine attacks within 48 h after discharge was 4 (30.8%) in the sodium valproate group, and nine (69.2%) in the ibuprofen group, indicating no statistically significant dif- ference (p = 0.147).
-
- Adverse events
Adverse events associated with the administered drugs were ob- served in two patients in the sodium valproate group (pain at the site of injection in one patient and dizziness in the other) and three patients in the ibuprofen group (nausea in two patients and dyspepsia in one pa- tient). Accordingly, the number of cases with adverse effects was similar between the groups. No systemic or life-threatening adverse event was observed in any of the patients.
- Discussion
In this study, our aim was to compare the analgesic efficacy of differ- ent medical treatments applied to patients who presented to the emer- gency department with an acute migraine attack. We administered sodium valproate and ibuprofen IV to our patients in two different treat- ment groups that we created. We then recorded the patients’ NRS scores at different measurement times. We compared the NRS scores obtained at different measurement times and the rate of changes in these scores from the baseline to each post-treatment evaluation time. We also eval- uated the patients’ requirement of rescue treatment during their stay in the emergency department and whether they had another migraine at- tack after discharge. As a result of our study, we found that the analgesic efficacy of IV sodium valproate was significantly higher than that of IV ibuprofen in the treatment of acute migraine attacks.
Headache due to an acute migraine attack is one of the common causes of emergency department presentations. The aim of treatment is to relieve pain, reduce the recurrence of attacks, and minimize the need for Rescue medication [17]. There is no standard treatment method that effectively and quickly relieves pain in migraine, and the response to treatment may vary from one patient to another. Non-specific treat- ment methods are generally used in the treatment of this type of head-
|
Ibuprofen (n = 50) |
Sodium valproate p value (n = 49) |
ache, especially in the emergency department. Ibuprofen is a first-line NSAID frequently used in acute migraine attacks with proven efficacy |
||
|
Age (years), mean Sex (female), n (%) |
35.1 +- 9.8 30 (60.0) |
34.3 +- 9.7 23 (46.9) |
0.816 0.193 |
[18]. By inhibiting Prostaglandin E2 synthesis, ibuprofen provides a di- rect effect on perivascular nociceptors in a migraine attack. In a previous |
|
BMI, mean |
26.3 +- 4.6 |
26.4 +- 5.1 |
0.978 |
study, Codispoti et al. showed that ibuprofen provided pain palliation |
|
Initial NRS score, mean |
8.9 +- 1.2 |
9.0+- 1.1 |
0.479 |
within two hours, even at low doses, and it was superior to the placebo |
Vital parameters
|
Systolic blood pressure, mmHg |
119.6 +- 14.4 |
120.4 +- 10.7 |
0.777 |
|
Diastolic blood pressure, mmHg |
70.6 +- 6.9 |
70.4 +- 7.1 |
0.892 |
|
Heart rate, bpm |
75.6 +- 13.1 |
72.4 +- 11.7 |
0.205 |
|
Oxygen saturation,% |
94.6 +- 2.7 |
94.1 +- 3.4 |
0.356 |
|
Respiratory rate, bpm |
13.8 +- 1.5 |
14.1 +- 1.9 |
0.391 |
|
Body temperature, ?C |
36.4 +- 0.4 |
36.5 +- 0.3 |
0.848 |
|
Pain Localization, n (%) |
|||
|
Unilateral |
41 (82.0) |
38 (77.6) |
0.581 |
|
Bilateral |
9 (18.0) |
11 (22.4) |
|
|
Duration > 24 hours, n (%) |
30 (60.0) |
21 (42.9) |
0.088 |
|
Attacks >= 4 per month, n (%) |
25 (50.0) |
32 (65.3) |
0.123 |
|
Nausea, n (%) |
45 (90.0) |
46 (93.9) |
0.479 |
|
Vomiting, n (%) |
23 (46.0) |
14 (28.6) |
0.073 |
|
Photophobia, n (%) |
35 (70.0) |
35 (71.4) |
0.876 |
|
Phonophobia, n (%) |
25 (50.0) |
27 (55.1) |
0.611 |
BMI; Body Mass Index, NRS; Numerical Rating Scale
in acute migraine attacks [19]. At the end of the two-hour period in our study, we found a clinically significant decrease in the pain scores of more than half of the patients that had been given ibuprofen. This rate is higher compared to the value reported by Kellstein et al., who exam- ined the efficacy of different doses of ibuprofen [20].
Sodium valproate is a medical agent that regulates cerebral excit- ability by acting as a central neurotransmitter [21], which is considered to be its mechanism of action in the treatment of migraine. In a clinical study, Shahien et al. reported that IV sodium valproate effectively relieved headaches associated with acute migraine attacks after a two-hour period [22]. However, clinical studies comparing the efficacy of sodium valproate with different drugs in acute migraine attacks were not able to reach a clear conclusion. In their clinical study, Fried- man et al. compared the efficacy of 3 different IV treatments (sodium valproate, metoclopramide and ketorolac) in patients presenting to
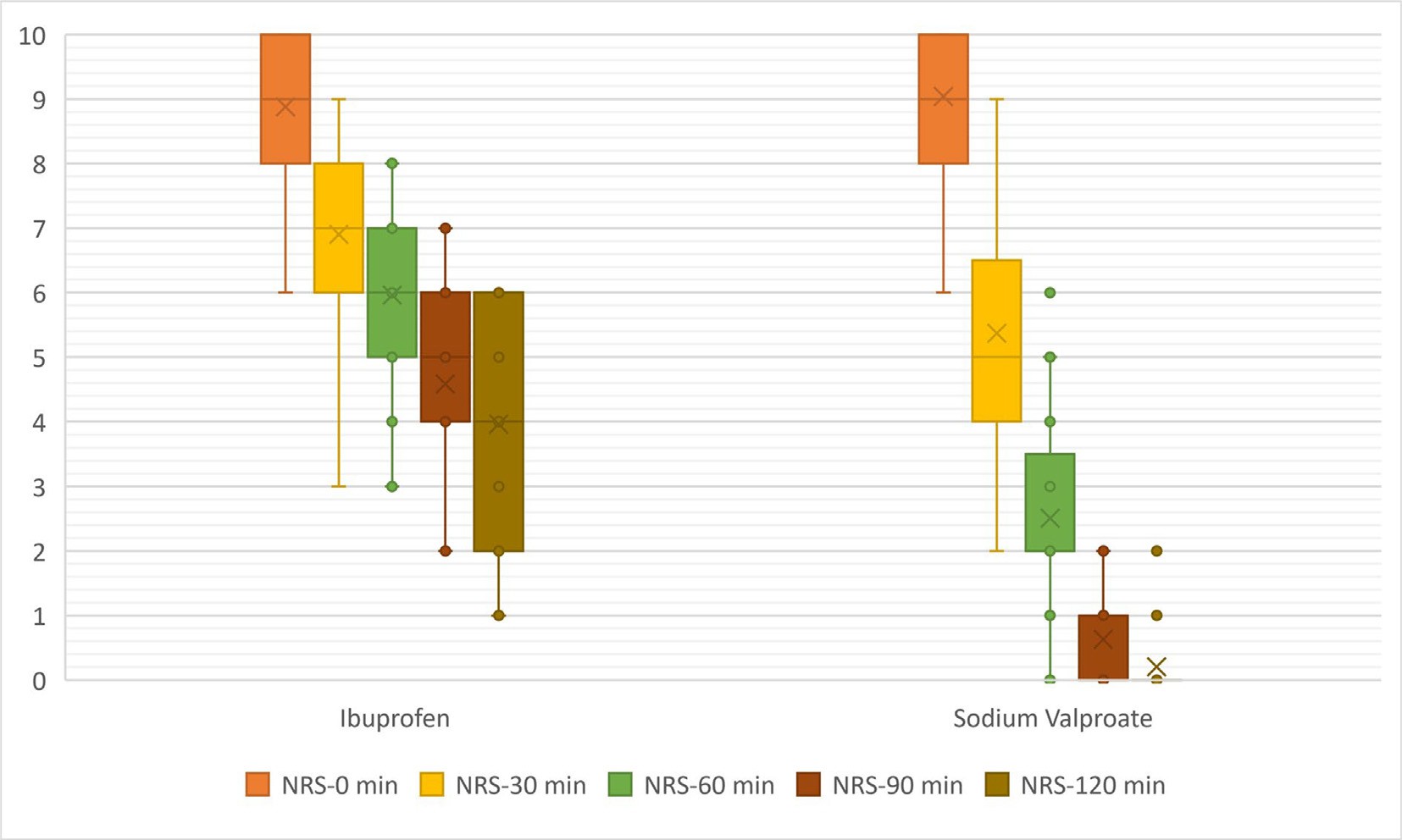
Fig. 2. Effect of sodium valproate and ibuprofen on the mean NRS scores measured at different times over the 120-min study period. NRS; Numerical Rating Scale, min; minutes.
the emergency department with acute migraine or acute probable mi- graine headache [16]. Results of that study indicated that patients re- ceiving valproate had a smaller improvement in pain score than patients receiving metoclopramide or ketorolac. However, data referred to as ‘mean improvement’ here are expressed as the mean pain score improvement over one hour, results at other measurement times and proportional reductions in pain scores are not stated. In this study by Friedman et al., there were 110 patients in each patient group, which
is considerably higher than the number of patients in our study. How- ever, in this literature study factors such as; not making a distinction be- tween migraine with and without aura, inclusion of Probable migraine headache patients in the study, not disclosing the BMI data of the pa- tient groups, and not showing ethnic group diversity may have caused different results about IV sodium valproate. In contrast, Rahimdel et al. found that IV sodium valproate and Subcutaneous sumatriptan had sim- ilar analgesic efficacy in acute migraine attacks [23]. Furthermore, in a
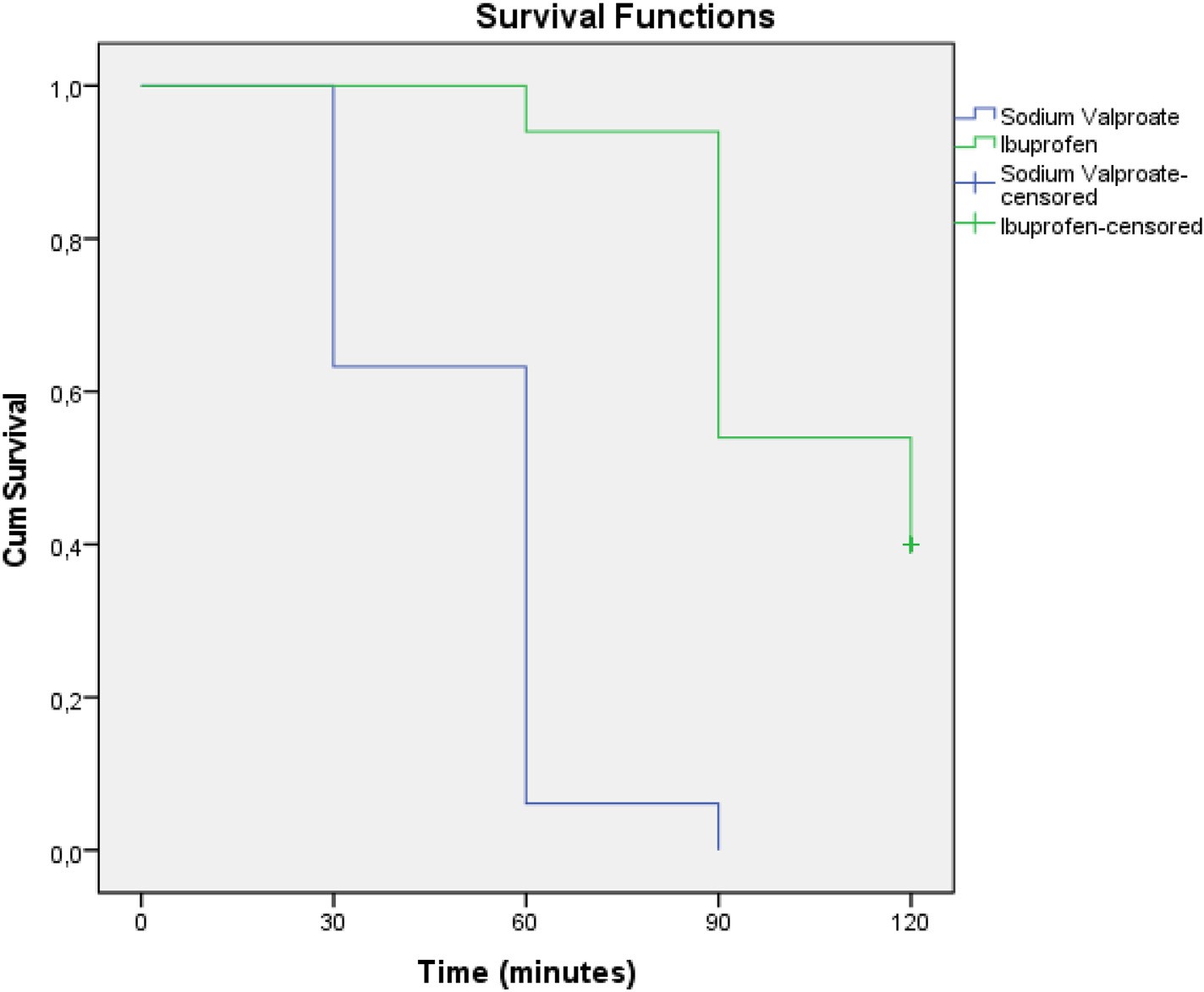
Fig. 3. The Kaplan-Meier curve showing the rate of reaching the endpoint at Numerical Rating Scale score measurement times according to the treatment groups.
study by Bakhshayesh et al., the efficacy of IV sodium valproate was de- termined to be higher than the co-administration of Intramuscular metoclopramide and subcutaneous sumatriptan [24]. These different results from clinical studies in the literature make it difficult to reach a consensus on the efficacy of sodium valproate. In the current study, we found that IV sodium valproate provided more effective pain relief than ibuprofen in the treatment of acute migraine.
One of the secondary endpoints of our study was a decrease in the frequency of migraine attacks. In this respect, we found no statistically significant difference between our treatment groups. However, the number of patients who had a recurrence within 48 h of discharge in the ibuprofen group was twice the number of those in the sodium valproate group. Saper et al. compared the rate of recurrence of acute migraine attacks within 24 h of treatment between the ibuprofen and Rofecoxib groups [25]. Similar to our study, the authors found that ibu- profen was not effective in reducing the frequency of attacks. In studies on the efficacy of sodium valproate in decreasing the frequency of mi- graine attacks, there was no clear evidence showing its superiority over another treatment (such as dexamethasone, metoclopramide, and ketorolac [16,26].
When we examined drug-related adverse events, we only observed non-systemic side effects in a small number of patients, which did not require the discontinuation of treatment. There was no difference be- tween the treatment groups in terms of the frequency of these adverse events. The adverse events and frequencies detected in our study were similar to those in previous studies in which both ibuprofen and sodium valproate were separately used in the treatment of acute migraine [2,27].
-
- Limitations
The main limitation of our study is that we excluded migraine pa- tients with aura from the sample. This is because our aim was to enroll patients presenting with migraine attacks in a single homogeneous group. Therefore, a study that evaluates the efficacy of ibuprofen and so- dium valproate in migraine patients can further contribute to the liter- ature. The second limitation of our study is that there were only two treatment groups and we did not have a placebo group. However, since ibuprofen and sodium valproate are drugs with previously proven efficacy and recommended in the treatment of acute migraine attacks, we considered that it was not necessary to create a placebo group in terms of clinical significance.
Based on the results we obtained in our study, sodium valproate can be taken into account in our daily practice as a new second-line treat- ment option that can be among the Analgesic drugs we are used to fre- quently using in emergency departments.
Declaration of interest statement”>Declaration of Interest statement
The authors declare no competing interests to disclose.
The authors declared that this study has received no financial sup- port or any funding.
CRediT authorship contribution statement
Sinem Dogruyol: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Sultan Tuna Akgol Gur: Conceptuali- zation, Formal analysis, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing. Ilker Akbas: Writing – review & editing, Writing – original draft, Resources, Project administra- tion, Investigation, Formal analysis, Data curation. Meryem Betos
Kocak: Conceptualization, Formal analysis, Methodology, Validation, Visualization, Writing – review & editing. Abdullah Osman Kocak: Writing – review & editing, Writing – original draft, Visualization, Vali- dation, Methodology, Formal analysis, Conceptualization. Mustafa Ceylan: Data curation, Formal analysis, Investigation, Project adminis- tration, Resources, Writing – original draft, Writing – review & editing. Davut Tekyol: Writing – review & editing, Validation, Supervision, Project administration, Methodology, Formal analysis.
References
- Orr SL, Friedman BW, Christie S, Minen MT, Bamford C, Kelley NE, et al. Management of Adults with Acute Migraine in the emergency department: the American head- ache society evidence assessment of parenteral pharmacotherapies. Headache. 2016;56(6):911-40. Epub 2016/06/15.
- Wang F, Zhang H, Wang L, Cao Y, He Q. Intravenous sodium valproate for acute mi- graine in the emergency department: a meta-analysis. Acta Neurol Scand. 2020;142 (6):521-30. Epub 2020/08/03.
- Steiner TJ, Stovner LJ, Vos T, Jensen R, Katsarava Z. Migraine is first cause of disability in under 50s: will health politicians now take notice? J headache pain. 2018;19(1):
17. Epub 2018/02/23. https://doi.org/10.1186/s10194-018-0846-2. PubMed PMID:
29468450; PubMed Central PMCID: PMCPMC5821623.
- Ashina M, Hansen JM, Do TP, Melo-Carrillo A, Burstein R, Moskowitz MA. Migraine and the trigeminovascular system-40 years and counting. Lancet Neurol. 2019;18 (8):795-804. Epub 2019/06/05. https://doi.org/10.1016/S1474-4422(19)30185-1. PubMed PMID: 31160203; PubMed Central PMCID: PMCPMC7164539.
- Ashina M. Migraine. N Engl J Med. 2020;383(19):1866-76. Epub 2020/11/20. https://doi.org/10.1056/NEJMra1915327. PubMed PMID: 33211930.
- Charles A. The pathophysiology of migraine: implications for clinical management. Lancet Neurol. 2018;17(2):174-82. Epub 2017/12/13. https://doi.org/10.1016/S14 74-4422(17)30435-0. PubMed PMID: 29229375.
- Tepper SJ, Spears RC. acute treatment of migraine. Neurol Clin. 2009;27(2): 417-27. Epub 2009/03/18. https://doi.org/10.1016/j.ncl.2008.11.008. PubMed PMID: 19289223.
- World Health Organization. Atlas of headache disorders and resources in the world 2011. Geneva: World Health Organisation; 2011..
- Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the american headache society evidence assessment of migraine pharmacother- apies. Headache. 2015;55(1):3-20. Epub 2015/01/21. https://doi.org/10.1111/ head.12499. PubMed PMID: 25600718.
- Rothrock JF. Clinical studies of valproate for migraine prophylaxis. Cephalalgia. 1997;17(2):81-3. Epub 1997/04/01. https://doi.org/10.1046/j.1468-2982.1997.1
702081.x. PubMed PMID: 9137842.
- Waberzinek G, Markova J, Mastik J. Safety and efficacy of intravenous sodium valproate in the treatment of acute migraine. Neuro Endocrinol Lett. 2007;28(1): 59-64. Epub 2007/02/06. PubMed PMID: 17277725.
- Karimi N, Tavakoli M, Charati JY, Shamsizade M. Single-dose intravenous sodium valproate (Depakine) versus dexamethasone for the treatment of Acute migraine headache: a double-blind randomized clinical trial. Clin Exp Emerg Med. 2017;4 (3):138-45. Epub 2017/10/14. 10.15441/ceem.16.199. PubMed PMID: 29026887;
PubMed Central PMCID: PMCPMC5635457.
- Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition. Cephalalgia. 2018;38
(1):1-211. Epub 2018/01/26. https://doi.org/10.1177/0333102417738202. PubMed PMID: 29368949.
- Bijur PE, Latimer CT, Gallagher EJ. Validation of a verbally administered numerical rating scale of acute pain for use in the emergency department. Acad Emerg Med. 2003;10(4):390-2. Epub 2003/04/03. https://doi.org/10.1111/j.1553-2712.2003. tb01355.x. PubMed PMID: 12670856.
- Akgol Gur ST, Dogruyol S, Kocak AO, Sanalp Menekse T, Akbas I, Betos Kocak M, et al. Topical capsaicin versus topical ibuprofen in Acute musculoskeletal injuries: a ran- domized, double-blind trial. Hong Kong J Emerg Med. 2020. 1024907920975368. ar- ticle in press.
- Friedman BW, Garber L, Yoon A, Solorzano C, Wollowitz A, Esses D, et al. Randomized trial of IV valproate vs metoclopramide vs ketorolac for acute migraine. Neurology. 2014;82(11):976-83. Epub 2014/02/14. https://doi.org/10.1212/WNL.00000000000
00223. PubMed PMID: 24523483.
- Krymchantowski AV. Acute treatment of migraine. Breaking the paradigm of mono- therapy. BMC Neurol. 2004;4(4). https://doi.org/10.1186/1471-2377-4-4. Epub 2004/03/10. PubMed PMID: 15005810; PubMed Central PMCID: PMCPMC341456.
- Suthisisang C, Poolsup N, Kittikulsuth W, Pudchakan P, Wiwatpanich P. Efficacy of low-dose ibuprofen in acute migraine treatment: systematic review and meta- analysis. Ann Pharmacother. 2007;41(11):1782-91. Epub 2007/09/20. https://doi. org/10.1345/aph.1K121. PubMed PMID: 17878396.
- Codispoti JR, Prior MJ, Fu M, Harte CM, Nelson EB. Efficacy of nonprescription doses of ibuprofen for treating migraine headache. A randomized controlled trial. Head- ache. 2001;41(7):665-79. Epub 2001/09/14. https://doi.org/10.1046/j.1526-4 610.2001.041007665.x. PubMed PMID: 11554954.
- Kellstein DE, Lipton RB, Geetha R, Koronkiewicz K, Evans FT, Stewart WF, et al. Eval- uation of a novel solubilized formulation of ibuprofen in the treatment of migraine headache: a randomized, double-blind, placebo-controlled, dose-ranging study. Cephalalgia. 2000;20(4):233-43. Epub 2000/09/22. https://doi.org/10.1046/j.1468- 2982.2000.00055.x. PubMed PMID: 10999673.
- Palermo A, Fierro B, Giglia G, Cosentino G, Puma AR, Brighina F. Modulation of visual cortex excitability in migraine with aura: effects of valproate therapy. Neurosci Lett. 2009;467(1):26-9. Epub 2009/10/06. https://doi.org/10.1016/j.neulet.2009.09.054. PubMed PMID: 19800389.
- Shahien R, Saleh SA, Bowirrat A. Intravenous sodium valproate aborts migraine headaches rapidly. Acta Neurol Scand. 2011;123(4):257-65. Epub 2010/06/24. https://doi.org/10.1111/j.1600-0404.2010.01394.x. PubMed PMID: 20569223.
- Rahimdel A, Mellat A, Zeinali A, Jafari E, Ayatollahi P. Comparison between intrave- nous sodium valproate and subcutaneous sumatriptan for treatment of acute mi- graine attacks; double-blind randomized clinical trial. Iran J Med Sci. 2014;39(2 Suppl):171-7. Epub 2014/04/23. PubMed PMID: 24753639; PubMed Central PMCID: PMCPMC3993044.
- Bakhshayesh B, Seyed Saadat SM, Rezania K, Hatamian H, Hossieninezhad M. A ran- domized open-label study of sodium valproate vs sumatriptan and metoclopramide
for prolonged migraine headache. Am J Emerg Med. 2013;31(3):540-4. Epub 2013/ 02/06. https://doi.org/10.1016/j.ajem.2012.10.014. PubMed PMID: 23380105.
- Saper J, Dahlof C, So Y, Tfelt-Hansen P, Malbecq W, Loeys T, et al. Rofecoxib in the acute treatment of migraine: a randomized controlled clinical trial. Headache. 2006;46(2):264-75. Epub 2006/02/24. https://doi.org/10.1111/j.1526-4610.200
6.00334.x. PubMed PMID: 16492236.
K. Randomized clinical trial of intravenous valproate (orifil) and dexamethasone in patients with migraine disorder. Iran J Med Sci. 2013;38(2 Suppl):150-5. Epub 2013/09/14. PubMed PMID: 24031104; PubMed Central PMCID: PMCPMC3771216.
- Karacabey S, Sanri E, Yalcinli S, Akoglu H. Which is more effective for the treatment of Acute migraine attack: dexketoprofen, ibuprofen or metoclopramide? Pak J Med Sci. 2018;34(2):418-23. Epub 2018/05/29. 10.12669/pjms.342.13815. PubMed PMID: 29805419; PubMed Central PMCID: PMCPMC5954390.
